
ACCELERATE TIME-TO-MARKET, IMPROVE OPERATIONAL EFFICIENCY
TrackWise® Quality
The industry standard in Quality Management – now with modules for Product Quality Review (PQR), Quality Management Review (QMR) and Recall Management
Achieve Proactive Quality, Improve Operational Efficiency & Effectiveness
For over 40 years, Honeywell has partnered with life sciences organizations to solve complex challenges and advance digital transformation. Our comprehensive portfolio – spanning quality, manufacturing, cybersecurity and analytics – is designed to improve operational efficiency, reduce risk and help bring safer products to patients faster.
Our integrated solutions support life sciences manufacturing operations of any size or complexity. We serve medical device and diagnostics manufacturers, pharmaceutical and biotechnology companies, and CDMOs across small molecules, proteins and peptides, cell and gene therapies, oligonucleotides and vaccines.
Smarter Quality. Seamless Operations.
End-to-End Solutions.
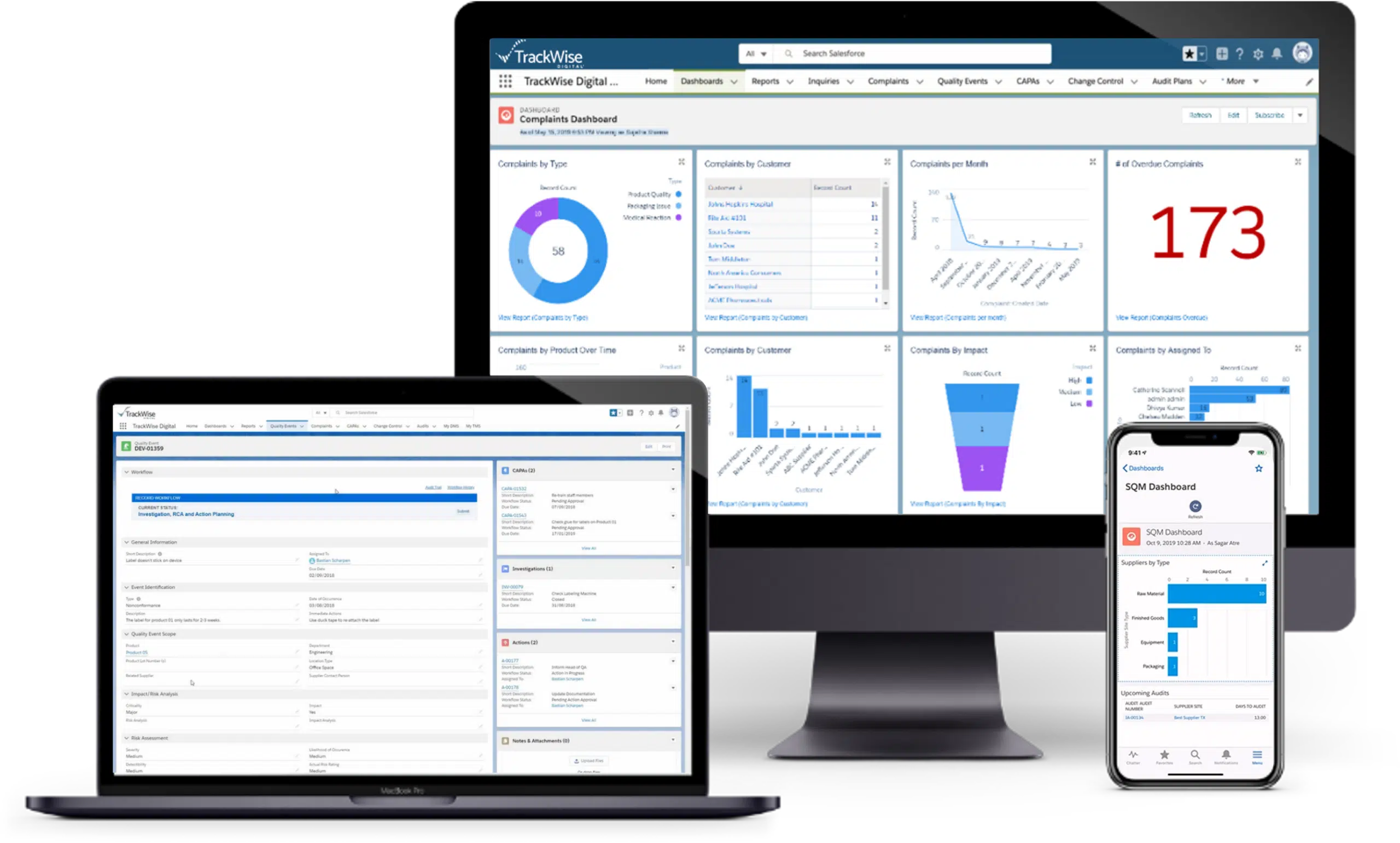
A Simpler, Smarter Quality Management System
Proactive quality depends on the ability to capture, connect and act on quality data. TrackWise Quality applies advanced technologies – including generative AI, natural language processing and advanced analytics – to help identify trends, manage quality events and reduce downstream risk. Gain real-time insights that support better decisions and enable earlier detection and resolution of issues before they escalate.
Everything You Need, in One Place
Establish a single source of truth for quality across the enterprise. From the shop floor to boardroom, TrackWise Quality helps teams monitor performance and streamline reporting through digital dashboards, optimized workflows and real-time visibility. Built-in QMR dashboards and PQR reporting tools support comprehensive oversight and clear communication of product and operational data.
Proven Expertise You Can Trust
Quality is foundational. Leading life sciences companies have relied on our quality solutions and domain expertise for decades to meet rigorous global standards and turn quality into a strategic advantage. Standardized, proven PQR workflows, help your processes meet global compliance standards while supporting continuous improvement and operational efficiency.
Faster Time-to-Value Through Harmonization
Simplify, standardize and scale quality processes with confidence. Whether implementing a new system or harmonizing quality globally, fit-for-purpose workflows grounded in industry best practices support GxP compliance and deliver faster realization of value.
Built for Regulatory Compliance
Meet evolving regulatory requirements with integrated, automated quality processes. TrackWise Quality maintains complete, accurate audit trails, supports inspections and helps reduce compliance risk – minimizing delays and disruptions to product delivery.
Proactive Risk Management
Protect product quality and patient safety with risk management capabilities aligned to ISO 14971 and ICH Q9. Built-in templates help teams identify, assess and mitigate risk earlier – reducing errors and supporting faster, more confident time-to-market.
Why Life Sciences Companies Trust TrackWise® Quality

Philips Healthcare
"It’s been a very collaborative partnership with Honeywell Life Sciences... and what was different with them is that they’re really helping us to develop the process that best works for us."
Steve C de Baca,
Chief Patient & Quality Officer

SGS
Digitalization of quality management across 17 global sites and larger partner network to improve operational efficiencies, capacity, turnaround times and customer service.
25K Documents
400 Audits
400 Audits

Insud Pharma
“We’re a company focused on health and well-being of, of women. Quality is at the heart of everything that we do. It’s built into the DNA of the organization.”
Ed Carey,
Global Health of Quality
Ed Carey,
Global Health of Quality

McKesson
"We’ve partnered with Honeywell Life Sciences and their TrackWise platform to help us build out a quality management system that’s designed and applicable for our space in life sciences."
Barry Cook,
VP of Quality Operation
VP of Quality Operation

Cipla
"I have seen the evolution of this application from version 6 to 9.5.1.1 and our company has actually rolled out 40+ workflows. This application has played a vital role in the successful clearance of multiple regulatory audits."
Srinivas Hosadurga, Senior Manager- IT

Olympus
"We are going through a process of standardizing, simplifying, and harmonizing the way we work, so that we can be efficient and compliant globally. And as part of that, we’re working with Honeywell Life Sciences."
Paul Belgrove, SVP of Corporate Quality
It’s Time to Modernize Your Quality System
No matter where you are in your quality management digital journey, Honeywell offers solutions to help you move forward with confidence.
TrackWise Digital®
TrackWise Digital is a modern, cloud-based quality management system with integrated modules that work together to support compliance, streamline quality processes and enable more efficient, data-driven decision-making.
TrackWise® QMS
TrackWise® is a proven, on-premise quality management system used by regulated life sciences organizations worldwide. It brings together industry best-practice workflows in a single system to help manage core quality processes consistently and effectively.
TrackWise AI
TrackWise AI delivers AI-enabled capabilities designed to enhance quality processes, support Good Manufacturing Practices (GMP) and help teams identify trends and insights more efficiently.
TrackWise® Quality Management Review (QMR)
The TrackWise QMR application modernizes the QMR process by reducing reliance on manual slide preparation and enabling automated data aggregation and analytics. The result is near real-time visibility with actionable, management-ready insights.
TrackWise® Product Quality Review (PQR)
The TrackWise PQR application streamlines the PQR process by providing a single digital workflow – from sourcing required data through draft report generation, multi-user review, approval, and final report output – supporting consistency, compliance and efficiency.
TrackWise® Recall Management
TrackWise Recall Management helps organizations reduce patient and business impact by providing a modern, digital recall solution that integrates with multiple systems of record to support faster, more coordinated response.
Key Insights & Resources for Smarter Quality Management
WHITEPAPER
Building A Business Case For QMS Investment
Do you have a compelling business case to support your QMS goals?
Whitepaper
Turning Quality Review Processes into Insights and Efficiencies through Business Applications
Frost & Sullivan Whitepaper
WHITEPAPER
Quality: The Link between Platform, Processes and Patients
In life sciences, effective quality management extends beyond compliance – helping organizations keep the patient at the center of every decision.
Webinar
Discover How Gedeon Richter Transformed Their Quality Management
Learn how leading European pharmaceutical and biotech company Gedeon Richter implemented a digital QMS to support business objectives and a patient-centric mission.
3 Steps To Digital-First Quality
Outdated or fragmented quality systems continue to challenge organizations of all sizes. This eBook outlines a practical three-step approach to adopting modern, cloud-based quality technology quickly and effectively.
WEBINAR
Revolutionizing Recall Management In Life Sciences
Recalls can affect public health, operational continuity and brand trust. Watch this webinar to explore modern approaches to managing product recalls more efficiently.













